|
To describe the reaction as it occurs we have to combine both mass preservation and molecules composition. It can be made when hydrogen reacts with oxygen - but these gases are usually present in the diatomic form of H 2 and O 2 molecules. 
A water molecule will always consist of one atom of oxygen and two atoms of hydrogen. We also know that compounds always have the same composition. The same happens with a charge - charge is also preserved, just like mass is. If the atom is present in a reactant (compound entering the reaction), it must be present in one of reaction products. We know that atoms don't appear nor disappear. Reaction balancing is based on mass preservation. Knowing the skeletal reaction equation we know what the reactants are and what are the products, but for quantitative predictions we need to balance the reaction equation. 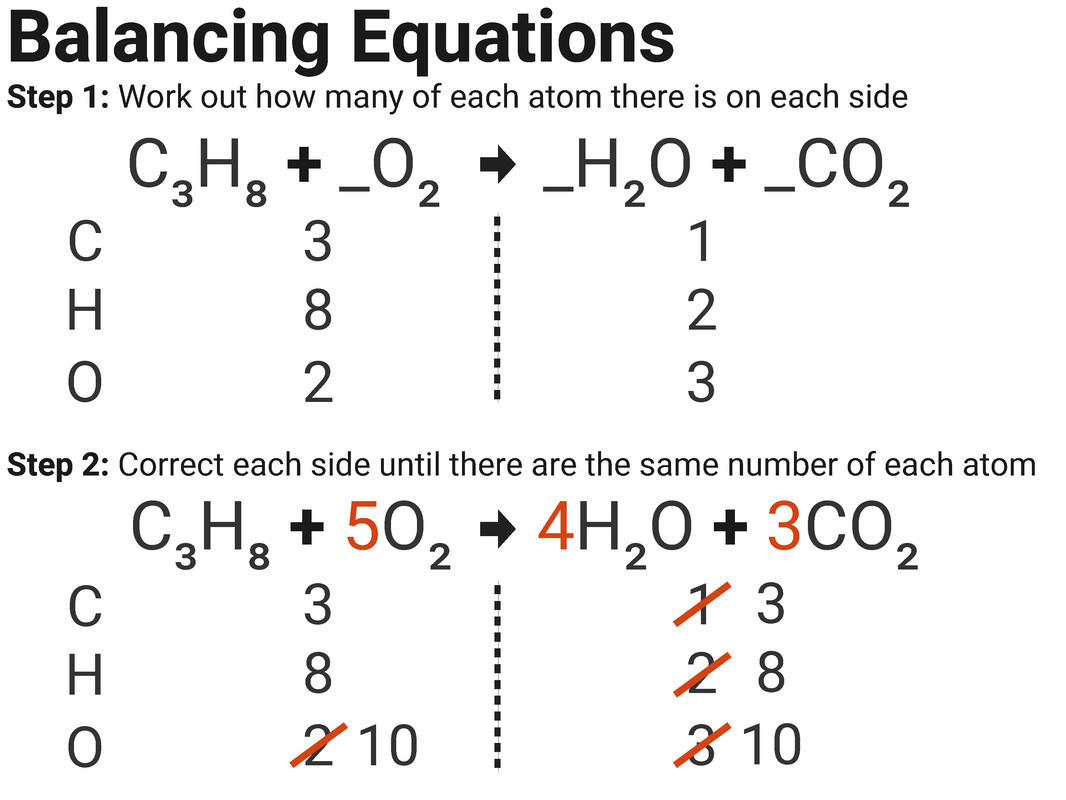
We call such a reaction equation (unbalanced, but correctly listing all reactants and products) skeletal. This reaction contains the correct reactants - hydrogen and oxygen in its diatomic forms, and correct product - water molecule. 
Why do we balance chemical reaction equationsĪll chemical reactions occurring within the test tubes, industrial reactors, or nature can be described by reaction equations.įor example reaction of water synthesis can be written as
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
